![]() 3.1 What is the dual stream model?
3.1 What is the dual stream model?
Given these separate anatomical accounts, attributing a function(s) to the arcuate is not clear cut, and any current account is far from the authoritative statement on the matter. Nonetheless, a vast majority of literature does place the arcuate as part of the dual stream model[1] of speech processing, although its exact role within these neural networks is still being disputed – and largely depends on which anatomical account you prescribe to.
The basic assumption of dual stream accounts is that phonological networks interact with both conceptual-semantic and motor-articulatory systems, leading to a distinction between the neural networks that process this speech information. These separate interactions are summarised under two processing streams: the dorsal stream and the ventral stream (Hickok and Poeppel, 2007). Connecting phonological networks with conceptual-semantic systems, using structures in the superior and middle portions of the temporal lobe, is the ventral stream. Meanwhile, the dorsal stream is linked via structures in the posterior frontal lobe to the posterior temporal lobe and parietal operculum, which connects phonological networks with motor-articulatory systems (ibid).
Phonological processing has two major aspects: receptive processing of phonemes and expressive production of phonemes (Glasser and Rilling, 2008). Current accounts of Wernicke’s area (posterior BA 22 and BA 40) place it as a hub for processing incoming phonemes (cf. Martin, 2003), while the production of phonemes takes place in the posterior portion of Broca’s area (BA 44 and BA 6) (cf. Bookheimer, 2002). Arguably, this notion of phonological processing can be considered the ventral stream, as described by Hickok and Poeppel (2007), and contrasts with the lexical-semantic system. Here, middle and inferior temporal areas (BA 21 and BA 37) connect with “[…] Broca’s area and frontal areas more anterior and superior to it (BA 44, 45, 47, 9)” (Glasser and Rilling, 2008, pg. 1). Besides lexical-semantic systems, these areas are also implicated in other higher functioning language domains, such as syntax (cf. Grodzinsky and Santi, 2008). This is the dorsal stream. Importantly, these two streams are disconnected from one another and are also left-lateralised.
As Hickok and Poeppel (2007) point out in their review, the dual stream model is far from a new idea, with Wernicke himself offering a similar proposal as part of his language model. Also, similar organisational systems are found in visual and somatosensory domains, which “[…] suggests that the present dual-stream proposal for cortical speech processing might be a reflection of a more general principle of sensory system organization.” (Hickok and Poeppel, 2007, pg. 393). This leaves us with working out the role of the arcuate within these networks, a task that will be discussed in the next section.
3.2 The dual stream model, the arcuate fasciculus and conduction aphasia
The classical incarnation of the arcuate is largely accepted as being involved in a dorsal-like processing system. Differences emerge in the extent of the arcuate’s anatomy, subsequently leading to different hypotheses for its functional role in speech processing. Of the literature read, it is perhaps suitable to frame these accounts by examining their explanatory power in the previously mentioned condition, conduction aphasia.
Conduction aphasia’s hallmark characteristic is difficulty in repetition, often accompanied by paraphasic impairments (phonological errors in fluency) and lexical-semantic deficits (difficulties in naming) (cf. Glasser and Rilling, 2008). As such, limiting the arcuate to a direct phonetic pathway reduces its ability to explain these cases, meaning it is specifically implicated in repetition problems – and disassociated from lexical-semantic impairments.
Catani et al. (2004) reconcile these characteristics by attributing them to lesions at different points in their extended perisylvian network, located on either the direct (phonetic) or indirect (semantic) pathways. Even though damage to the traditional, direct route for the arcuate is reduced to just explaining repetition, the indirect pathways extend into areas associated with lexical-semantic functions. Lesions to these extended pathways explain the differences observed in some patients. Nonetheless, this does not explain the observations of Hickok and Poepell (2004), who argue conduction aphasia is more likely to result from cortical dysfunction, explained by damage to “[…] the sound-based speech processing systems in left STG and/or to the temporal–parietal system (Spt) which interfaces these systems with motor–articulatory networks.” (ibid, pg. 93).
Taking these considerations in mind, Saur et al. (2008) further relegate the arcuate and do not attribute it any special status within the dorsal stream. Instead, they speculate that phonological impairments of conduction aphasia are the result of a disruption in their proposed dorsal arcuate/SLF system. This offers a strong explanatory case for why conduction aphasia does not always result from an arcuate lesion. It also does not rely on extending the perisylvian network and fits in with additional DTI studies of the arcuate’s anatomy (Schmahmann et al., 2007; Frey et al., 2008). Here, lexical-semantic impairments are not explained, which is probably because they offer a different anatomical route for their ventral stream, which uses the extreme capsule (EmC) to connect temporal and ventrolateral prefrontal cortex; and therefore, it is presumed the authors likely ascribe to an account similar to those presented by Hickok and Poepell (2007), Dronkers et al. (2007) and Schmahmann et al. (2007).
Offering another position is Glasser and Rilling (2008), who see the arcuate as a critical substrate of both ventral and dorsal pathways, including right hemispheric language processing (see figure three). This version places it somewhat at odds with the previous studies mentioned, as they claim the left-lateralised arcuate is not a direct phonetic pathway but two functionally distinct neural networks. Importantly, Wernicke-like and Broca-like conduction aphasias are explained under an adapted version of Hickok and Poepell’s (2004) model, with lesions in the STG pathway causing phonological impairments, whilst damage to the MTG pathway is associated with lexical-semantic impairments.
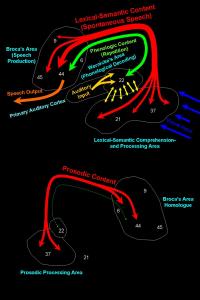
Furthermore, they also acknowledge the EmC acting as a ventral pathway, which accounts for instances where the arcuate lesion does not cause conduction aphasia or where conduction aphasia arises from lesions in different regions (Glasser and Rilling, 2008). This leads them to conclude that:
“[…] conduction aphasia can be best explained by damage to the relatively superficial STG and extreme capsule pathways and/or the surrounding phonologic cortex, rather than a deeper lesion that damages mostly the more medial MTG pathway of the arcuate fasciculus, as argued by the classic model.” (ibid, pg. 7).
Still, this account does not necessarily negate alternative explanations for conduction aphasia, nor does it definitively place the arcuate as a critical component of the dual stream – processing both ventral and dorsal streams. Further work is needed before these pathways can be fully elucidated and subsequently attributed a functional role. However, given evidence of recent selection pressures (Rilling et al., 2008) – something neither the SLF nor EmC show signs of – the arcuate seems likely to be subserving a critical role in processing of a human-related behaviour, language being a plausible candidate.
4. Conclusion
Discerning the role of the arcuate fasiculus in speech processing is a process yet to be completed. This understanding might evolve out of current models based on speech processing, especially the dual stream model discussed above, which seems to provide an intuitive, comprehensive understanding of the mechanisms involved. Based on DTI tractography, we can now begin to appreciate the complexity of the arcuate, and at the same time realise that these approaches are not all revealing — as demonstrated in the differences between studies, where some show pathway terminations in more frontal regions (Rilling et al., 2008), whilst others claim these connections are non-existant (Saur et al., 2008).
That these differing interpretations exist does not necessarily hinder all attempts to ascertain a functional role for the arcuate substrate. For instance, it is reasonable to assume that, under the dual-stream model, the arcuate is almost certainly involved in mapping phonological networks with motor-articulatory systems. What remains to be solved is whether or not this pathway also extends into conceptual semantic domains i.e. the ventral stream. To elucidate upon this, we need to not only enhance the range of DTI studies performed, but to also offer more comparative accounts between primates, other mammals and the neurological regions/networks themselves.
Still, the dual stream model, for all its explanatory capacity, is unsubstantiated. And, even if the dual stream account is largely accurate, the arcuate is not necessarily a critical component, and may just be part of several substrates connecting language-associated regions. This leads us to another viable criticism: the apparent connections between function and anatomy are not always clear-cut. As such, one might argue the regions described are involved in other cognitively demanding processing — see: tool use, action sequences and other complex behaviours. Despite these possible avenues, recent insights into the arcuate’s anatomical, functional and evolutionary underpinnings suggest a functional role within speech processing. Whether this role underpins both dorsal and ventral pathways remains an open question.
[1] Catani and Mesulam (2008) actually prescribe to McCarthy and Warrington’s two-route model of speech production, which, as you have probably already guessed, focuses soley on speech production.
Main References
Hickok, G. (2004). Dorsal and ventral streams: a framework for understanding aspects of the functional anatomy of language Cognition, 92 (1-2), 67-99 DOI: 10.1016/j.cognition.2003.10.011
Glasser, M., & Rilling, J. (2008). DTI Tractography of the Human Brain’s Language Pathways Cerebral Cortex, 18 (11), 2471-2482 DOI: 10.1093/cercor/bhn011
Schmahmann, J., Pandya, D., Wang, R., Dai, G., D’Arceuil, H., de Crespigny, A., & Wedeen, V. (2007). Association fibre pathways of the brain: parallel observations from diffusion spectrum imaging and autoradiography Brain, 130 (3), 630-653 DOI: 10.1093/brain/awl359
Excellent review highlighting the yet undeveloped areas of research. For another review article on the topic including other white matter structures see:
Friederici, A.D. (2009). Pathways to language: fiber tracts in the human brain. Trends in Cognitive Sciences Vol.13 (4)
Thanks Yisrael. I’ve just downloaded Friederici’s paper, and at a first glance it seems like a good read.